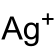重铬酸银
| 重铬酸银 | |||
|---|---|---|---|

| |||
| |||
| IUPAC名 Silver dichromate | |||
| 识别 | |||
| CAS号 | 7784-02-3 | ||
| PubChem | 9932493 | ||
| SMILES |
| ||
| InChI |
| ||
| 性质 | |||
| 化学式 | Ag2Cr2O7 | ||
| 摩尔质量 | 431.76 g·mol⁻¹ | ||
| 外观 | 深红色粉末 | ||
| 密度 | 4.77 g/cm3 | ||
| 溶解性(水) | Ksp = 2.0×10-7 | ||
| 若非注明,所有数据均出自标准状态(25 ℃,100 kPa)下。 | |||
重铬酸银是无机化合物,化学式为Ag2Cr2O7,不溶于水,遇热水分解。
制备
[编辑]- K2Cr2O7 + AgNO3 → Ag2Cr2O7 + 2KNO3
应用
[编辑]相关的配合物在有机化学中用作氧化剂。[1]例如,重铬酸四吡啶合银,[Ag2(py)4]2+[Cr2O7]2-,用作将苄醇和烯丙醇类化合物转化为相应的羰基化合物。[2]
参考资料
[编辑]- ^ Firouzabadi, H.; Seddighi, M.; Ahmadi, Z. Arab; Sardarian, A. R. Selective Oxidative Cleavage of Benzylic Carbon-Nitrogen Double Bonds Under Non-Aqueous Condition with Tetrakis(pyridine)-Silver Dichromate [(Py)2Ag]2Cr2O7. Synthetic Communications. 1989, 19 (19): 3385. doi:10.1080/00397918908052745.
- ^ Firouzabadi, H.; Sardarian, A.; Gharibi, H. Tetrakis (Pyridine)silver Dichromate Py4Ag2Cr207 - A Mild and Efficient Reagent for the Conversion of Benzylic and Allylic Alcohols to Their Corresponding Carbonyl Compounds. Synthetic Communications. 1984, 14: 89. doi:10.1080/00397918408060869.
- Greenwood, N. N.; Earnshaw, A. Chemistry of the Elements 2nd. Oxford:Butterworth-Heinemann. 1997. ISBN 0-7506-3365-4.
| 这是一篇关于无机化合物的小作品。您可以通过编辑或修订扩充其内容。 |